In recent years, Platelet Rich Fibrin (PRF) has gained immense attention and popularity within the dental community for its regenerative capabilities. This technique enhances the body’s natural healing process, accelerates tissue repair, and promotes optimal healing in dental surgeries. Dentists worldwide are embracing this cutting-edge approach, recognizing its potential to improve wound healing and patient outcomes.
What is PRF?
Platelet rich fibrin (PRF) is a technique that utilizes the patient’s own blood components to stimulate natural repair processes. This speeds up healing after a dental surgery. A centrifuge separates red blood cells from other blood components in a small collected blood sample, deriving PRF as a usable fibrin clot packed with multiple healing and growth factors.
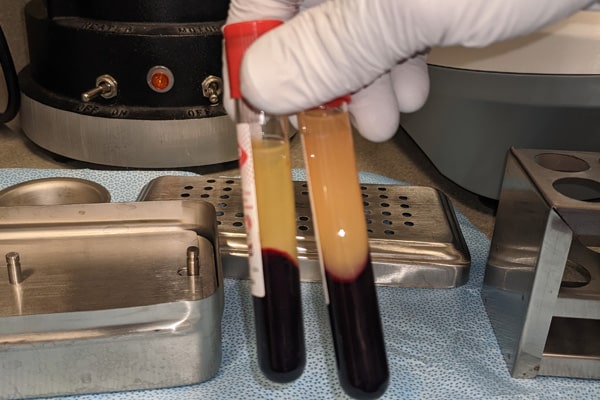
The fibrin clot, a concentrated matrix of fibrin, is enriched with platelets, white blood cells, growth factors, and cytokines. The supporting fibrin matrix may also enhance the body’s ability to fight infection1https://pubmed.ncbi.nlm.nih.gov/21740376/ which would prevent some complications from various dental surgical procedures.
Origins and evolution of PRF
Dr. Joseph Choukroun and his colleagues originated platelet rich fibrin (PRF) in 2001. They were searching for an alternative to Platelet Rich Plasma (PRP), leading to the discovery and development of PRF, a second-generation platelet concentrate.
Over time, PRF evolved and gained traction in the dental community due to its numerous advantages. PRF treatments have diversified and evolved to improve the density of the platelets, growth factors and supporting fibrin matrix. Varying the centrifugation process allows the use of different layers for various healing processes. From this, various types of platelet rich fibrin techniques such as platelet rich fibrin – Leukocytes (PRF-L) and Injectable platelet rich fibrin (i-PRF)2https://pubmed.ncbi.nlm.nih.gov/23189893/3https://pubmed.ncbi.nlm.nih.gov/25806786/ were developed, which are all aimed to improve patient outcomes.
Differences Between PRF, PRP, and Other Platelet Concentrates
Platelet rich fibrin, Platelet Rich Plasma, and other platelet concentrates like injectable platelet rich fibrin (i-PRF), and platelet rich fibrin – leukocytes (PRF-L) all originate from the same source, but they differ in their properties and applications.
Comparing PRF and PRP
Platelet rich plasma is the earliest predecessor which is used for its high concentration of platelets.
These platelets have a high growth factor concentration. This results in accelerated tissue healing and bone regeneration with platelet rich plasma injections. However, platelet rich plasma has a short lifespan, thus limiting its effectiveness over prolonged periods. Additionally, platelet rich plasma requires anticoagulants which may negatively impact the healing wound.
Platelet rich fibrin works a little differently. Unlike platelet rich plasma, PRF also utilizes fibrin, a protein that aids in clotting. The fibrin network resorbs slower which allows for a slow, sustained release of growth factors. This provides a scaffold for cells to adhere to which makes PRF especially effective in regenerative procedures. Furthermore, PRF doesn’t use anticoagulants, making it a great graft material.
Injectable PRF (i-PRF) and PRF-Leukocytes (PRF-L) are derived from the PRF technique. i-PRF remains in a liquid form for a longer period which allows for it to be injected into place, similar to a PRP injection. It also can be blended with bone graft materials. It blends the best of both platelet rich fibrin and platelet rich plasma. PRF-L, on the other hand, includes a higher concentration of the white blood cell called leukocytes. These white blood cells fight bacteria which can reduce the changes of infection after the procedure4National Library of Medicine. (2020). “Biological Foundation and Clinical Application of Platelet Rich Fibrin in Dentistry.” Journal of Dental Medicine..
Advantages of PRF over other concentrates
While platelet rich plasma, i-PRF, and PRF-L all hold their respective advantages, PRF stands in many dental surgical procedures. This is due to the fibrin matrix supporting a sustained growth factor release leading to better patient outcomes. This unique attribute makes PRF an excellent choice for dental procedures requiring tissue or periodontal regeneration. This provides long-lasting benefits to the patients.
PRF in Dentistry: Production and Composition
All forms of this technique, regardless of being platelet rich plasma or PRF, all start off with a simple and safe blood draw. Usually, a few vials of blood (10-20ml) is drawn from a vein, like with a routine blood draw. A tourniquet is placed and blood is drawn usually from the inside of the elbow. Drawing and using the patients’ own blood results in a material that is biocompatible. It also carries little to no risk of immune reactions or disease transmission.
How is Platelet Rich Fibrin Produced?
After the blood draw, the vials filled with blood are placed in a centrifuge. A centrifuge is a machine that spins the blood sample at high speeds. This separates its components based on their densities. Typically, the centrifugation process takes 10 to 12 minutes. This is done at various speeds, which may range from 2,000 to 5,000 rotations per minute (rpm). Variations in RPMs and spin time produce different products, including PRP injections, i-PRF, and of course PRF.
Biological Composition of PRF
Since each product is made from the same blood, the composition of each product is similar. For PRF, the spinning process separates the blood into three layers: a layer of red blood cells, a layer with the PRF clot, and another layer of acellular plasma. The PRF clot looks like a semi-transparent gel. It is then retrieved and often placed in various types of presses to be shaped for use in surgical procedures, even as a resorbable membrane.
This PRF clot contains a concentrated blend of platelets, leukocytes, and growth factors. These components all play a crucial role in wound healing and tissue regeneration. These components are powerful tools for cell signaling which improve outcomes in many dental surgical procedures such as various bone grafting and periodontal regeneration5https://www.tandfonline.com/.
Once clinicians isolate the PRF clot, they can mold it into membranes or plugs suitable for grafting techniques. These PRF products not only foster quicker healing but also reduce post-operative complications. As an autologous material, PRF minimizes the risk of allergic reactions and disease transmission. Its applications in dentistry, backed by extensive research, underscore its significance in improving treatment outcomes and enhancing patient care3https://pubmed.ncbi.nlm.nih.gov/25806786/.
Mechanism of Action of PRF in Dental Procedures
Platelet rich fibrin utilizes an enriched fibrin framework, capable of releasing growth factors and cytokines. These components stimulate cellular proliferation and angiogenesis, resulting in faster and more efficient healing6https://www.ncbi.nlm.nih.gov/pmc/articles/PMC4509294/. PFT also helps regulate the inflammatory response which can limit the swelling, reduce pain and improve the postoperative experience, comfort and satisfaction7https://pubmed.ncbi.nlm.nih.gov/27783851/. Lastly the leukocytes are known to minimize infection rates. This is due to the role white blood cells play in the human immune system.
PRF and tissue regeneration in the oral cavity
PRF excels at soft tissue regeneration, which has great application in surgical dental procedures such as extractions. A key concern for both patients and dentists is the reduction of post-operative complications such as dry socket, also known as alveolar osteitis. PRF treatment has been shown decrease post operative complications and discomfort8https://www.ncbi.nlm.nih.gov/pmc/articles/PMC5529189/ and can even help treat dry socket which is a common complication9https://www.ncbi.nlm.nih.gov/pmc/articles/PMC8060184/. Moreover, PRF’s coagulative properties can reduce post-operative bleeding, which is especially beneficial for patients with coagulation disorders or taking anticoagulants.
Benefits of Using PRF in Dental Treatments
Incorporating PRF into dental procedures improves wound healing, reduces postoperative discomfort, and results in better treatment outcomes. The material, taken from the patient, minimizes the risk of biocompatibility issues and disease transmission, providing a cost-effective, practical solution for enhanced patient treatment success.
Clinical Applications of PRF in Dentistry
Many dental surgeries will benefit from the use of PRF. Most of the applications involve surgeries, both soft and hard tissue surgeries. PRF is not helpful in achieving better results with a dental crown or compositeA synthetic resin that is used for while fillings. This is what most people receive when they get fillings. fillings due to the non-surgical nature. Several specialties that can utilize PRF include implant dentistry, periodontics, and oral and maxillofacial surgery.
PRF, used in guided bone regeneration (GBR) or sinus lifts, releases growth factors to encourage bone maturation.. In the case of tooth extractions, PRF reduces the occurrence of dry socket. It can also reduce post operative pain, and hastens soft tissue healing, ultimately facilitating faster recovery. There are many benefits to the usage of PRF, with few downsides which result in it’s increasing popularity.

Research and Studies Supporting PRF Efficacy in Dentistry
There is a large body of evidence that supports the effectiveness of PRF in improving patient outcomes. Additional studies to the ones previously mentioned include a 2020 study headed by Dr. Richard Miron in the Journal of Oral Implantology. This meta-study reinforces the effects of PRF in the recovery of bone defects10https://www.sciencedirect.com/science/article/pii/S1121417115000187. Additionally, there are some studies that have shown that PRF can reduce post operative pain and improve bone healing in surgical root canal procedures11https://pubmed.ncbi.nlm.nih.gov/21740376/. All these findings support the usage of PRF in dental procedures due to its ability to improve patient outcomes.
Possible Side Effects and Risks of PRF in Dentistry
There are little to no side effects or risks when using PRF in dentistry, however there are some potential risks. The blood draw itself, not the PRF procedure, can have some complications such as a hematoma, infection, and fainting. Other risks involve contamination or incorrect handling which may affect the quality and efficacy of PRF itself. However, following sterile protocols greatly reduces these risks. Since the blood used is autogenous, meaning it comes from the patient, there is virtually no risk of rejection. Due to these, there are significant benefits with few risks associated with PRF.
The Future of PRF in Dental Medicine
PRF will continue to be an effective addition to many dental procedures. New innovations with PRF will revolve around establishing new protocols to produce different products. A few examples are the leukocyte and injectable variations. Customization of PRF products will enable clinicians to tailor treatments based on specific patient needs. This will allow for more specific uses for variation in PRF, which will all improve healing and regeneration. Further personalizing treatment modalities will lead to better patient outcomes and better oral and systemic health.
Was this post helpful?
Table of Contents
- What is PRF?
- Differences Between PRF, PRP, and Other Platelet Concentrates
- PRF in Dentistry: Production and Composition
- Mechanism of Action of PRF in Dental Procedures
- Benefits of Using PRF in Dental Treatments
- Clinical Applications of PRF in Dentistry
- Research and Studies Supporting PRF Efficacy in Dentistry
- Possible Side Effects and Risks of PRF in Dentistry
- The Future of PRF in Dental Medicine
References
- 1
- 2
- 3
- 4National Library of Medicine. (2020). “Biological Foundation and Clinical Application of Platelet Rich Fibrin in Dentistry.” Journal of Dental Medicine.
- 5
- 6
- 7
- 8
- 9
- 10
- 11